Readers Summary
- Is delaying aging a better strategy than preventing cancer, heart disease and obesity for wellness?
- What are the 3 major effects of oxidation in biology?
- Is water a giant molecular three-dimensional adaptor for sunlight?
- How does the power in the sun get to glutathione via water?
- Why is our rarest amino acid, cysteine, so incredibly important?
Let’s start out with an intro; watch this video before proceeding further.
In my opinion, quantum theory predicts the future evolutionary path clinical medicine will take. The road to Optimal has an alliteration using the “letter P” associated with it. QED will make medicine preventative, predictive, personalized, participative, very soon in my humble opinion. The question remains, can the world’s people wait that long for the evoluted change to occur?
Today’s blog ideas are a beginning of bringing that vision to life for many. I plan on continuing to teach those who are interested in learning this new clinical and evolutionary path. That vision is buried in a pathway in us where wellness morphs into illness and aging.
So, what is the most important signaling pathway in all life forms for longevity and for optimal health? The short is the answer is the redox potential that is stored in the membranes, organelles and the exclusion zone of water in cells. I will be bold to say that the maintenance of intracellular redox homeostasis is the most important thing to get right if you want to get optimal.
The pathologic effects of oxidative stress can be funneled down to three major situations for cells:
- Lipid Peroxidation of plasma and cell membranes
- Protein oxidation causing destruction or aggregation of proteins
- DNA/RNA oxidation causing mutation and chromosomal instability
How we are designed to optimize this pathway is very complex. The key factors are based upon is the interaction of water and thiol groups in an amino acid called cysteine.
The sun generates massive energies across the entire electromagnetic spectrum. Water stores those energies. Water is the conduit for the electromagnetic spectrum to act through the electromagnetic force. This quantum connection allows for molecular three-dimensional recognition without any close contact required. This process happens via electromagnetic waves and their native resonant frequencies.
This is how energy is naturally transferred to biology. The electromagnetic force can store an infinite range of potential energies delivered to it from the electromagnetic energies from the spectrum of light from the sun. The potential energy is transferred from the native electromagnetic spectrum via the electromagnetic force to water molecules in cells. The potential energy is stored in water is then transferred to the sulfur-containing thiol groups of cysteine. This means water is a bidirectional molecule designed for all types of energy transfers.
Fundamentally, water is a chameleon. It is globally elastic and locally fragile because it can act in many different forms it can assume chemically and electrically. Water can organize itself differently when it is adjacent to different materials, allowing it to be the perfect chemical for interactions with the electromagnetic force.
Think of water as the ultimate molecular photoelectric adapter for life. Water’s behavior is a function of its microenvironment. An MRI is proof of this concept. Water looks different in different tissues of the human body, yet, it still remains H20 at a biochemical level of observation. This biochemical scale is not good enough to explain how life organizes. To understand how life makes use of water, you need a smaller scale: a quantum scale.
Water energy transfers are governed by something called “bandgap engineering.” This is a quantum mechanical action found in chemicals/proteins that are capable of semiconduction. Becker found this exact same mechanism in bone biology when he realized that two copper ions were the doping mechanism that rectified the photoelectric effect in the periosteum of bone to transfer bone’s photoelectric signal into a mechanical one for bone apposition and resorption.
Light and water also do the same thing as proteins like cysteine. It is the “potential” of the thiol groups in cysteine where your redox potential really lies. Cysteine is the rarest amino acid in nature, and this fact alone makes it an ideal signaling molecule in redox biochemistry. It links aging and neolithic disease risk, and it also marks where wellness begins and ends in us all.
The redox of the thiols in cysteine
Now that you have a basic idea of how a quantum cell works (see the November 2013 webinar), let’s discuss how this cell interacts with the first biologic protein force I mentioned in EE 11, the redox reactions in the Pentose Phosphate Pathway and beta-oxidation of fats.
If you go back and reread EMF4, you will see that one of the major benefits of living within that pathway is to make NADPH that replenishes glutathione. This pathway has only a few major endpoints, but they are all massively important for human life. It is designed to optimally restore RNA and DNA synthesis by providing maximum recycling of ATP; you now know the real physiologic function of ATP is to withdraw electrons from proteins’ electron clouds to cause them to fully expand and open its binding sites to water. This is Gilbert Ling’s work; he proved it all experimentally, yet no one listened to him.
The PPP also allows for the optimal formation of bio-energetic substrates to replenish the mitochondria’s ability to increase total adenine nucleotides to re-establish optimal ATP stores from beta-oxidation of fats. This is why we make 147 ATP from beta-oxidation of fats and only 36 ATP from carbohydrate metabolism. This difference is not just a calculation of ATP stoichiometry, it is a function of the electromagnetic spectrum energies during the seasons present on Earth as they change. Therefore, the electromagnetic spectrum of the sun carries information and energies of these seasonal factors directly to mitochondria. Mitochondrial proteins are used to decipher this “Seasonal Rosetta Stone” of energies.
This is a critical point between the two major arms of metabolism. Mitochondrial cytochrome proteins are like MRI machines for electrons and protons. They are designed to tell the difference in the energies of electrons and protons energies and information. From those two physical differences, the proteins within the cytochromes dictate the pathway where those subatomic particles should flow.
When we are in an environment that is withdrawing energy from us, while causing entropy to rise, our cells and tissues want all the metabolic and hormone help we can muster to remain well. This implies that metabolism and hormone levels are optimized when our ability to separate charges in water is maximized.
Another way to state this in metabolic biologic jargon: When carbohydrates are used consistently 24/7 in long light cycles, this implies cytokines levels remain high (warmer); therefore, inflammation is high, proton levels are highest for mitochondria to handle, and glutathione levels inside a cell are very low. These physical and electrical differences lead to effects in cellular proteins and water to alter the molecular spin (known as Larmor frequency) of the hydration cells around your nucleic acids, proteins, membranes, and around your mitochondria:
The molecular spins around RNA and DNA are also drastically altered daily and seasonally due to the sensitivity and specificity of the mitochondria and its proteins. This directly alters your epigenetic programming and its library’s variability and ability to react to all environmental pressures from changes in the sun’s electromagnetic spectrum of energies and forces in our environment. These electrical changes are measured in your redox potential in every cell of your body. The direct effects are found experimentally in DNA methylation and histone acetylation patterns in cells.
When glutathione drops, it implies that you are using up your cysteine stores too quickly. It turns out using up cysteine too fast makes you age faster than your biologic age and makes you ill on the way to accomplishing that task. It also implies thiamine stores are destroyed as redox power is lost. As a result, pyruvate rises and so will blood lactate as ATP drops.
The PPP has one more key element that many have forgotten: it makes and restores the key reducing chemical element, NADPH, to replenish glutathione in all cells, but especially the liver. This is why detoxification of any toxin is directly linked to the redox potential of any cell. Clearing a toxin does you no good if you cannot generate a redox potential. I mentioned that in EMF 4. REDOX BEFORE YOU DETOX!!!!
Glutathione is the major antioxidant in the body and is a critical element for reducing inflammation, oxidation, and detoxification in the body. To enter it routinely, it requires the human to be able to accurately tell proper “quantum time” in your cytochromes, SCN and your liver. This is why all life has both central circadian clocks and organ clocks. This is an alteration of “quantum time” is often seen in the abnormal secretion of pepsin and betaine function in the gut. You’ll see evidence of pseudohypoxia in cells as thiamine drops and more dysautonomia present. When this cycle is off, your gut hormones will be off as well.
If your endogenous molecular clock is off, you can bet you are now concentrating transition metals in your body’s tissues. This happens because methionine, glutathione and picolinic acid cycles are all off. Thiamine links to all these pathways as the mitochondrial matrix fails. A failing matrix is another term for heteroplasmy. This pathway will stay in your blind spot and you will continue to believe you need carbohydrates to replenish glycogen post-exercise to make ATP or replenish glutathione from fructose. The act of “concentrating transition metals” has a massive effect on energy production by lowering it in the quantum cell. We will cover this special situation in its own blog to discuss aging in finer detail.
How does this connect to Leaky Gut?
For severe prolonged leaky gut or autoimmune conditions, IV glutathione treatments hold a lot of promise. You would be wiser to add food high in cysteine to your diet because glutathione supplements are difficult to get into cells by any route compared to your gut. It also requires optimal B12, folate and betaine levels when treatment is ongoing.
To optimize these naturally from food requires proper circadian signals from cortisol and melatonin within the brain. Both of these rely deftly on Vitamin A and D cycles in the brain and body, respectively. When a person is more oxidized, there is a shift in our immune systems’ T helper cells, favoring the TH1 arm of the immune system over the TH2 system. Eventually, this mismatch develops into most of the autoimmune diseases you all have heard of. TH1-dominated responses are involved in the pathogenesis of organ-specific autoimmune disorders, Crohn’s disease, sarcoidosis, acute kidney allograft rejection, and some unexplained recurrent abortions.
Both oral and IV use of NAC or glutathione also uses up zinc as a cofactor, so I usually recommend zinc supplementation when it is used. N Acetyl Cysteine is the precursor molecule for glutathione, which is the main antioxidant protectant of our body. People with serious skin manifestations of the leaky gut like psoriasis, acne, or eosinophilic folliculitis should consider NAC because it directly blocks IL-4 which is important in the TH2 immune activation. This, in turn, is the main factor in producing IgE antibodies. This is a huge issue for vegans. IgE antibodies are made in hay fever, asthma, anaphylactic shock, and atopic skin diseases.
Skin conditions walk hand and hand with gut and light/dark abnormalities in the SCN. They are directly tied to poor Vitamin A recycling in the brain. This is tied to the photoperiod that a cell senses from its environment. As a result, these people with skin issues, also have serious alterations in the mechanism that controls the photoperiod in the brain controlled by vitamin A. This is why dermatologists find Retin A drugs so useful for skin conditions. If asthma is severe, one can measure the amount of nitric oxide directly in the expired air and it will be elevated. All neuroimmune diseases have these results at some level, as I laid out in Energy and Epigenetics 8. This is also true in cases of emphysema, COPD and cystic fibrosis. (Corradi et al. 2001)
The NAC will form glutathione, and it directly combines with the nitric oxide to create nitrosothiols (these thiol groups are going to be a big deal later in this blog). This binding reduces the inflammatory effects of the nitric oxide in the body quickly to limit disease. This is also critical within the intestines and manifests as a “leaky gut.” A leaky gut is really synonymous with a serious glutathione issue or a poor redox potential. This ties directly back to an altered light cycle because of its relationship to cortisol and/or melatonin or the Vitamin A and D cycles.
What I am describing here to you is how the electromagnetic signal is transduced from light, encoded in the serotonin stores in your gut lining, which signal the pineal to release melatonin for sleep when light is absent by way of the directional spin of CSF vortex, to glutathione levels in every tissue which directly controls the flow of electrons on the inner mitochondrial membrane to gain wellness or cause disease. Food is broken down to electrons, and water is charge-separated into oxygen-containing molecules and protons. In this sense, you begin to realize that hormones are the electronic quantum biologic signal of the electrical redox potential in the cell.
Food is not designed to be a metabolic fuel. It is a hormone information that tells you about your redox potential and its charge. If your redox potential is good, you will have the ability to handle most foods. When it is not, you will need to change your environment and diet to one that matches your redox potential best.
This is where most people find themselves as they live their life today, yet do not realize it. Sometimes changing your diet is not enough. The redox potential is 100% linked to the environment you find yourself in. When the information in your hormones is lacking (negative entropy), this ruins your ability to perform electron and proton transfers in your mitochondria’s cytochromes, as you will soon find out.
GLUTATHIONE is a small protein composed of just three amino acids: cysteine, glutamic acid and glycine. This becomes important because one-third of glutathione is made from cysteine. It is a semi-essential amino acid, which means that it can be biosynthesized in humans from methionine. Although classified as a non-essential amino acid as it used to be thought in rare clinical situations, cysteine may be essential for those with poor immune function (like infants and the elderly) and individuals with certain metabolic disease or who suffer from malabsorption syndromes. Today, however, because the environment has radically changed in the last 63 years, many more people have a huge need for cysteine/cytosine.
Glutathione is critical in maintaining optimal redox reactions, like the Fenton reactions you learned about in Energy and Epigenetics 8. Optimizing your intracellular glutathione levels is how you combat aging and neolithic disease generation best, in my opinion. Glutathione cannot get rid of the hydroxyl free radicals from the Fenton reactions, but the hydroxyl free radicals cause other proteins and metabolic pathways to become oxidized, and glutathione can help limit that collateral damage. As glutathione drops it acts as a proxy for a failing thiamine balance in tissues. When the process is excessive, glutathione levels inside a cell will be runway down, and your immune function will also seriously decline and open you up to many other disease processes.
This is precisely what happens with T2D, chronic fatigue, mold illnesses, Lyme disease, autoimmune conditions, and fibromyalgia. Glutathione recycling becomes the battle line of how the quantum cell maintains its ability to stay chemically reduced for as long as possible.
Humans tend to become more oxidized and less reduced as they age overall normally. The rate at which you become oxidized tells you how early you will succumb to neolithic disease and how fast you will age. When glutathione function goes awry, it allows biologic aging to become uncoupled from your chronological age. This shortens your telomeres and your life span shrinks, as does your healthspan. Clinically, you also feel terrible. Many people with poor glutathione recycling will feel an impending sense of doom.
Glutathione is produced normally by the body in response to today’s environment, reacting to conditions such as pollution loaded with transition metals in the air we breathe or the water we drink; or heavy metals, chemicals & pesticides in the food we eat (which damages our cells & system). What is often not appreciated is how glutathione is tied to energy generation and detoxification to maximally decrease the entropy in a cell.
A decrease in entropy means two things in the quantum world:
- It means less chaos and more information is present in the system. Anything that oxidizes our cells causes us to make less oxygen from our mitochondria for respiration. This creates molecular chaos and swelling inside a cell. The cell uses these changes to adapt to stresses. The key signalers are RNS and ROS.
- It also decreases the exclusion zone that water can generate. When this happens, our tissues begin to slowly collect transition metals like aluminum, zinc and heavy metals in our cytosol. This is analogous to what happens in a star just before it dies as a supernova.
A decrease in entropy is a quantum effect, not a biologic one, contrary to popular belief, mirroring how nature acts fundamentally, in your body and in a star. When the redox potential is lost, all matter struggles to deal with transition metals. Glutathione recycling is how these reactive transition metals are controlled in life. Sulfur-containing compounds also control the condensation of gases that form galaxies in space. The congruence of nature is astounding if you examine it.
Chelating these transition metals is often a very bad thing to do when your cellular cytosol has simultaneous low levels of cysteine or glutathione. Freeing up a toxin when you cannot clear it makes the situation worse, not better. Very few people get told that by healthcare professionals. When chelation is done in the face of low glutathione or cysteine levels, massive levels of inflammation can occur very quickly and this accelerates aging and disease processes.
Vegans have very low levels of cysteine by design of their diet, and their tissues made of collagen show this effect first. One of the first places a human can assess their glutathione functioning is by looking in the mirror at their skin. If you have a lot of skin infections or red erythematous skin for any reason at all, you can bet your intracellular glutathione levels are not close to optimal. Glutathione in increased doses posts a remarkable good “side effect,” such as good SKIN coloring. The gut and lung are the next places you will see the effect.
The greater the exposure to any toxins, the faster the body uses up its glutathione to detoxify other chemicals and metals in the body. This is why so many people with “gut issues” simultaneously have poor skin. People also forget that there is a brain gut axis where cysteine and glutathione interactions become critical. The skin and brain are also derived from neuro-ectoderm, so if your skin is suboptimal, you can bet your brain is also inflamed to some degree. Here is where the Vitamin A and D cycles are impacted because of how they are fundamentally tied to the electromagnetic spectrum of light.
Without the protection of intracellular glutathione, cells die at a faster rate, making people age quicker and putting them at risk for toxin-induced diseases like cancer.
How cysteine makes glutathione
Cysteine can usually be synthesized by the human body under normal physiological conditions if a sufficient quantity of methionine is available. Cysteine is the rarest amino acid and there is a deep evolutionary reason glutathione is made from a lot of this specific amino acid. It ties back to much of the work done by Gilbert Ling, as you will soon see. Very few foods have cysteine in them. Cysteine is found in most high-protein foods, including:
- Animal sources: pork, poultry, eggs
- Plant sources: red peppers, garlic, onions, broccoli, brussels sprouts,
People on Okinawa tend to eat a lot of pork, seafood, garlic, onions and peppers, and they live a long life. Some in the paleosphere will tell you Okinawans eat a lot of carbs. This is just not reality. The Okinawan diet uses ideal combinations of foods to help them remain very chemically reduced. Many of these foods are contained in my EPCOTx treatment protocol, which in part helps address degenerative disc disease and gut issues (have a look back at the Leaky Gut Rx for a review).
So why did evolution specifically pick cysteine as the best “Maxwell Molecular Demon” when it is the most rare amino acid? If you are rare in nature, it means you can be used as a great signaling molecule when you are present or absent. The levels of cysteine becomes something the “coherently linked system” uses to couple many other metabolic reactions. Signaling begins and ends with the precise molecular array of this amino acid. In this way, the cell can link energy balance to entropy generation in the entire organism.
So, how does a cell do this?
What is the primary biological functions of cysteine?
Here is where Gilbert Ling’s physical chemistry work begins to make its huge impact. Glutathione acts as a non-enzymatic reducing agent to help keep cysteine thiol side chains in a reduced state on the surface of proteins. The longer they remain reduced, the longer they evade protein clearance by the ubiquitin system. Why is this critical? Protein synthesis in cells costs the cell the most energy. It is such an energy-dense process that is can deplete cells and tissues of stored electrons faster than any molecular process in the body.
If you can use your proteins longer and do not need to replace them so often, you preserve your stem cells. The more times you invoke protein renewal, the more you have to tap your stem cells, and you age and die faster. Glutathione is involved in the synthesis and repair of DNA and RNA. It also assists in recycling peripheral antioxidants, Vitamin E and C. It inhibits free radical damage while enhancing the antioxidant activity of Vitamin C. Glutathione also facilitates the transport of amino acids and plays a critical role in the detoxification of metals and oxidative chemicals.
Glutathione is the base substrate for several other key antioxidant enzyme systems, such as glutathione-peroxidase, glutathione-reductase, and glutathione-transferase. Moreover, declines in cysteine and glutathione concentrations in intracellular fluids correlate directly with indicators of aging found in cells. This directly links aging and neolithic diseases to the redox potential of cysteine. A poorly-working redox system pushes a cell from being metastable and toward equilibrium (death state) and makes it more chaotic and be less energy efficient because proteins that are oxidized are not fully expanded to bind to water properly. Remember, Ling said when a cell dies is the only time it truly is at equilibrium.
When water binds in this fashion, it can form a larger exclusion zone (EZ) to generate a larger amounts of charge separation. The larger the EZ is, the more energy is delivered within the entire organism to remain metastable.
Biologic Aging for the Biology Geeks
It appears as we age, cysteine levels change dramatically first. It appears that cysteine is the major regulator of biologic aging inside a cell because of how it affects the glutathione cycle and our ultimate redox potential. When it falls, we cannot separate water into its positive and negative components. GSH is reduced glutathione and GSSH is called oxidized glutathione. The oxidative stress of aging is due to a deficiency in the rarest amino acid, cysteine.
The literature is now littered with many articles about this linkage. It is time you realize this. It appears cysteine deficiency is directly tied to epigenetic activation or deactivation via its coupling or decoupling to the NRF2 pathway. This happens when a cell mitochondria are not making enough water. Nrf2 stands for the Nuclear factor (erythroid-derived 2)-like 2. NRF2 and its downstream target genes are the primary cellular defense against the cytotoxic effects of oxidative stress.
The NRF2 pathway is directly tied to inflammatory cytokine formation and activation and increases in entropy. Keap1-Nrf2 pathway is of particular importance to this rare amino acid because of how Keap 1 and cysteine are linked. Keap 1 has four discrete protein domains in which cysteine residues are critically important in stress, sensing of oxidation. This cysteine binding element also is where the NRF2 pathway binds directly to Keap 1. This shows you why cysteine sits in the most critical spot for oxidative sensing in all human cells.
KEY POINT: The higher oxidation potential manifests as an extracellular decrease in the ratio of cysteine/cystine and an intracellular decrease in the ratio of GSH/GSSG and NAD(P)H/NAD(P). This implies that a metabolic shift in the redox state of the cell is more important than ROS made in the mitochondria or on macromolecules. So lower levels of cysteine and cystine are seen in aging and disease as a first signal of where disease begins and accelerates the aging process.
BIOLOGY GEEKS: Here is some of the complexity I am trying to leave out for the non-geeks. The Keap1-Nrf2 pathway plays a crucial role in determining the sensitivity of cells of the ABC transporters. ABC transporters are ATP-binding cassette transporters are members of a protein superfamily that is one of the largest and most ancient families with representatives in all extant phyla from prokaryotes to humans. ABC transporters are transmembrane proteins that utilize the electrons of adenosine triphosphate (ATP) and GTP to fully unfold biological proteins involved in the translocation of various substrates across membranes and non-transport-related processes such as translation of RNA and DNA repair. Although most eukaryotic ABC transporters are effluxes, some are not directly involved in transporting substrates.
For example, both the cystic fibrosis transmembrane regulator (CFTR) and the sulfonylurea receptor (SUR) are directly affected by ATP’s ability to cause a full open confirmation of its proteins for proper action. This is associated with the regulation of opening and closing of “ion channels demon proteins” using GTP. These proteins are affected by ATP and GTP levels to directly affect the protein conformational folding of the human ABC protein. Here again, we see ATP/GTP being used as an electron-withdrawing cardinal absorbent to open the protein conformations of these proteins in membranes. This implies the more ATP we make, the more accurate these transporters will function in cellular signaling. This is why a high-fat diet is beneficial for longevity and health. What food gurus forget is 2/3 of ATP in a human is made when that human is out in the sunlight that contains UV-A and IR-A light. Why? UV-A light makes nitric oxide which shuts down tunneling of electrons from cytochrome 1, 2, 3. Food electrons enter mitochondria there. Sunlight makes ATP at cytochrome c oxidase by turning off proximal ECT and using the 42% of IR-A light in sunlight. Sunlight provides double the amount of ATP production that food can provide. We seem to forget this. The more ATP made, the accuracy of cellular signaling, the less chance you age or get ill. This is why sunlight trumps food. Considering quantum cell theory, it really is that simple.

NON GEEKS: Anti-oxidant response element up-regulation, unfolded protein response, mitochondrial biogenesis, DNA/RNA repair mechanisms, and autophagy are all things that lead to wellness and reduce aging. These are all completed when cysteine and glutathione cycles are working optimally via the PPP. The PPP is important for making optimal amounts of ATP from the mitochondria.
Optimal maintenance of redox homeostasis ensures proper circadian cycles are in effect. When these become uncoupled for any reason at all, you are getting ill and not staying well. These cycles are of primordial importance for optimizing energy utilization and minimizing entropy, implying that glutathione cycling is directly tied to extreme low frequency EMF signaling as we laid out in EE 8. People do not understand how food and EMF impacts the epigenetic expression of RNA and DNA.
All redox reactions in the body are tied at some level to the photoelectric effect, water, and the electromagnetic force.
Glutathione production in cells links the photoelectric effect in foods. When we are oxidized, we use up our stores of cysteine and glutathione. Excessive inflammation, from any cause, does not allow us to replenish its levels and it does not allow us to separate water into its positive and negative charges and create large exclusion zones. This limits energy production in a cell while slowly degrading cell signaling.
All these coupled metabolic pathways use proteins that use transition metals as catalysts to lower the energy of activation of all coupled metabolic pathways. Coupling of pathways is done by proteins that act like Maxwell Demons to minimize energy loss and keep entropy low. This means that cysteine has to be included in the RNA and DNA motifs of these “molecular demons proteins” to signal the redox status. This explains why glutathione is loaded with cysteine in its 3D molecular structure for this specific reason. These facts should make it clear how important redox reactions are in wellness and in aging.
All health or illness conditions, lifestyle choices, and environmental conditions can accelerate the loss of glutathione or cysteine and alter the oxidative shift in the redox state. This information is sent to our mitochondria and it affects how we handle the subatomic particles in foods and from water.
Epigenetic “Oxidative Redox Shift” Theory of Wellness and Aging
Harman’s free radical theory of aging posits that oxidized macromolecules accumulate with age to decrease function and shorten lifespan. However, nutritional and genetic interventions to boost antioxidants have generally failed to increase life-span. When an experiment does not match your theory, your theory is wrong.
This is why the use of dietary antioxidants is now felt to be very problematic. Just a few years ago, everyone was telling people to eat more foods with high ORAC levels. This is a bad idea because our redox potential is mostly manufactured by charge separation and proper energy transfers and not 100% tied to our food intrinsic qualities. Furthermore, Harman’s free radical theory has failed to explain why exercise causes higher levels of oxygen radical damage but generally promotes healthy aging. This paradox can be fully explained using Quantum Cell Theory.
In order to maintain resting energy levels and minimize molecular chaos, aerobic glycolysis has to be upregulated by redox-sensitive transcription factors. It has been established that defects in mitochondrial respiration are not the ultimate cause of cancer or aerobic glycolysis (Warburg effect), thus the advantages of enhanced glycolysis in cancer remains quite controversial today.
Many cells ranging from microbes to lymphocytes use aerobic glycolysis during rapid proliferation in cell growth, which suggests it may play a fundamental role in supporting cell growth. As we laid out in EE 7, aerobic glycolysis is linked to the MHC1 gene that gives humans their superior longevity compared to primates. Aerobic glycolysis is critical in providing carbon atoms to generate biomass for normal or abnormal growth. This is tied to the development of the human immune system from the MHC 1 gene.
Normal aging with longevity requires optimal growth in the neuro-immune system cells. Cancer and autoimmunity seem to “steal this pathway” when it needs to hoard carbon to drive its own growth. I believe it happens by de-coupling the MHC 1 gene from the normal redox control system of glutathione and cysteine.
This is why autoimmunity always seems tied to a “leaky gut.” The number one cause of a leaky gut is non-native electromagnetic forces not allowing you to generate sufficiently-charged particles in your tissues to increase your redox potential. Food is a secondary player in this equation. This concept is paradoxical when you do not see how quantum effects act upon proteins and water collectively.
Food is an additive, not primordial, cause–in most cases. It is due directly to a severe lack of recycled cysteine and glutathione in the cytosol of your quantum cell. This means a lack of cysteine causes glutathione to no longer be able to act as a perfect Maxwell demon in cells, and people become energy inefficient and they lose energy to their environment. This causes you to begin to concentrate on transition metals that induce aging via a change in a process called ubiquination. This change increases entropy in our immune cells. This directly links glutathione and cysteine to healthy aging and cancer.
This is only a paradox to biology, medicine, and biochemistry because they do not appreciate how nature really operates at a quantum level. The “paradox” is only a conflict between reality and your feeling of what reality “ought to be.” (EPR paradox becomes important because it is a quantum effect.)
Consider that researchers have realized that if we can cure cancer, we may be able to also improve lifespan by three years based on recent studies. Sadly, no one is close to solving that equation because the answer is not based on the gene, it is based on how the conductor makes the music from the genes!
A lower level of glutathione also impairs tertiary and quaternary protein folding. This becomes critical in neurodegenerative disorders and in antibody formation, another link to autoimmune diseases.
MITOCHONDRIAL BIOLOGY GEEKS: Cytochrome 1 is where most ROS is made normally in humans. This is important in mitochondrial signaling and in biogenesis. This is where NADH and NAD+ act, and where the carbon atoms of carbohydrates feed into the mitochondrial complex. Proton movement is also important inside mitochondria. We have to move the protons in our mitochondria created to harvest their energy, or we decrease the size of the exclusion zone of water, which limits our redox potential.
It also turns out the need to supply NAD+ for glucose oxidation is needed to maintain optimal redox balance. When cytochrome 1 is impaired, this cannot be done. So the task falls to the other cytochromes to make up the deficit. Cytochromes are also called oxidoreductases. In biochemistry, an oxidoreductase is an enzyme that catalyzes the transfer of electrons from one molecule, the reductant (also called the electron donor) to another oxidant (also called the electron acceptor). When we speak of electron transfer we are speaking of quantum tunneling of these electrons through the molecular Maxwell demon proteins embedded in these cytochromes. This group of enzymes usually utilizes NADP or NAD+ as cofactors to speed the process up. This is where the lost NAD+, from cytochrome one, is made, to rebalance a loss of reductive power. This implies that mitochondria respond by making more ROS offset the loss of NAD+. All inflammatory signals carry a positive charge in biochemistry.
Riddle me this: why would Lady Evolution use positively charged molecules to signal? Could it be that the background resting transmembrane potential (that Ling said was built by electronic induction and not a membrane pump) be NEGATIVELY CHARGED? The short answer is, you can bet your ass that is why she uses a positively charged subatomic particle such as the proton to signal within the mitochondria.
This signal is generated at complex one. This is not something modern biochemists fully accept, but the data is becoming more clear that this is how it works in reality. To make sense of the paradox mentioned above, we need to realize the redox state is the bio-energetic force that allows for quantum electron tunneling to happen in mitochondria.
The charge across this inner mitochondrial membrane is massive. It has been shown that this change across this membrane causes a pH gradient as well as an electrical charge of about 150 millivolts across the inner mitochondrial membrane. This may sound like a small amount of charge, but consider this: the inner membrane is only 4-5 nanometers thick, so the voltage across this membrane is about 30 million volts per meter! For comparison, that is equivalent to the energy in a bolt of lightning. It is an electrical gradient that drives electrons from food to reduce oxygen from our lungs.
This is what powers all life. How this charge is electrically induced is the key to life. Ling is the guy who found out how it was done and still satisfied the Second Law of Thermodynamics. It uses quantum physics, specifically quantum tunneling at mitochondrial complexes. The way electrons enter the “quantum tunneling chain” is the key to the paradox. The truth does not cease to exist when it is ignored. Reality is that which, when you stop believing in it, doesn’t go away.
KEY LEARNING POINT: Nature is a riddle wrapped in a mystery rolled into an enigma and buried in paradox. Just as the light of a candle casts a shadow of darkness, sometimes the keys to the gates of heaven also open the gates of hell. The opposite of a correct statement is a false statement. But the opposite of a profound truth may well be another profound truth. This is the essence of paradox and shows is the counterintuitive basis of nature at its core.
When a cell is oxidized, its pH is low, and pH is a measure of the strength of proton transfer to make ATP. This is how the inner mitochondrial membrane is built by evolution to also be a “Maxwell Molecular Demon” to not only make energy but also to also limit molecular chaos. When you have this “quantized insight” about the function of the inner mitochondrial membrane and its cytochromes, it makes sense that “metabolically initiated redox shift” is found “upstream” of the commonly observed increase in ROS damage to macromolecules.
This is something you would expect a quantum cell to do, and not one based upon modern cell theory. The fact that many researchers are now finding it experimentally to be true should tell modern biology they need to look deeper or ask better questions. This metabolic shift worsens normally as we age, and it is not just seen in disease states. We lose our ability to “chemically reduce” as we age. It also means we become more oxidized into the direction of the relative levels of reductants like glutathione and cysteine within the cell. This manifests as an extracellular decrease in the ratio of cysteine/cystine.
We also find an intracellular decrease in the ratio of GSH/GSSG and NAD(P)H/NAD(P). This is why glutathione and NADPH fall in unison. This is why you begin to perceive you need carbs and remain sedentary (see EMF 4). When cytochrome 1 is just 2% inefficient for any reason, NAD+ must be made somehow to stimulate mitochondria for biogenesis. Without mitochondrial biogenesis (PQQ), you age faster and your organs fail quicker. An oxidative shift would ensure ample supplies of the requisite NAD+. This is why quantum cells react as they do.
When this happens chronically in humans, oxidation drives mitochondrial signals that lead to early mitochondrial replacement and a loss of mitochondrial capacity. This is called Quantum Aging due to a loss of charge across your mitochondrial membrane. The membrane is fine … but the charge separation is not well at all. This explains why disease and aging look exactly the same when we examine it.
| Cellular Phenomena | Cancer Cells | Senescent Cells |
|---|---|---|
| Inflammation | upregulated | upregulated |
| Apoptosis | apoptosis resistant | apoptosis resistant |
| Telomeres | short | short Inflammation |
| Inflammation | high | high |
| Mitochondria | damaged | damaged |
| Reactive oxygen species | high levels | high levels |
| Antioxidant enzymes upregulated | yes | yes |
| Inflammatory cytokine secretion | yes – IL-6 driven | yes – IL-6 driven |
| (SASP) Immune evasion | yes –TGF-β driven | yes – TGF-β driven |
| Defective DNA repair | yes | yes |
| DNA mutations | present | present |
| Harmful epigenetic silencing | yes | yes |
| Harmful epigenetic desilencing | yes | yes |
| mTOR upregulated | yes | yes |
| Ras/Raf pathway upregulated | yes | yes |
| c-myc pathway upregulated | yes | yes |
| Angiogenesis upregulated | yes | yes |
| Extracellular matrix degradation (big) | upregulated | upregulated |
| Sex steroids | down-regulated | down-regulated |
This point is not a small one. It is massively important and ties directly into the information I hope you read in the last blog, Energy and Epigenetics 11.
The electromagnetic force controls the action of all charged particles with infinite power. It turns out that all cellular flows and reactions in cells have been reflexively ascribed to thermally induced density gradients as a dogmatic belief. I am showing you in the last blog and this blog that this is simply a belief and not true. Charged based forces are much greater than thermally induced forces or gravitational forces.
To appreciate the difference, consider a proton and electron situated next to one another: Which natural force dominates their electrostatic interaction? Most people would guess the electromagnetic force but what would shock them is: the magnitude of the attraction of the electromagnetic force is on the order of 10 to the 38th power stronger! When we have this level of power at play, you the patient need to understand that small charge gradients on the inner mitochondrial membrane can lead to massive flows of electrons and protons in mitochondria. This can be good in the case of health or it can be bad in the case of disease or aging.
NON-GEEKS: This means we are designed to grow old, and as we grow old we also get ill. As we lose our ability to remain metastable, we lose our ability to act in concert, to be coherent in maintaining the charges in our mitochondria. To be as sharp as a tack, and to be fit as a fiddle, these two aspects are yoked in us by evolutionary design. I told you in the Quantum Sleep blog the purpose of sleep was to keep us metastable and not at equilibrium.
Remember, being metastable is the way life dances with the Second Law of Thermodynamics. It turns out The Second Law of Thermodynamics defines quantum time’s arrow in the evolution of all things living. This captures what we know to be true from our experience. As time elapses, we age. As a cookie crumbles, it cannot be reassembled. When we scramble eggs, we can not make them whole uncracked eggs once again.
But, Mother Nature has a curveball for us in QED. The laws of quantum physics are time reversible: one cannot derive a time’s arrow from them, nor are they affected by a reversal of time. This means if we can stay metastable and far from equilibrium as we live, life and wellness can be extended for a long period of time, much longer than we expect for humans even now. This is really good news for us all, and better news if you are sick. The application of how to do this is part and parcel of what I have been sharing with you for three years now.
The actions mentioned above, in mitochondria, act to shorten a cell’s life on purpose; this allows organs to age faster and simultaneously, stem cells are depleted. This redox signal is required to get the conductor to make some “new music” in the form of mitochondrial biogenesis.
What is that new epigenetic music? To avoid this catastrophic cycle, lactate dehydrogenase becomes epigenetically up-regulated at the expense of lactic acid acidosis. This metabolic shift becomes epigenetically enforced by histone deacteylation, as discussed briefly in EE8. It also turns out insulin resistance manifests as well, epigenetically, as discussed in EE 11. This occurs quickly to reduce mitochondrial turnover.
When this metabolic shift happens, it immediately lowers the mitochondrial capacity for efficient production of energy. It is a protective shift, not a pathologic one. It is adaptive. This lowers the current of flow of electrons from food, slows electron tunneling, and lowers eventual ATP production. It also affects how protons are handled by our mitochondria as well.
Lower ATP production does not allow water to bind to proteins to form large exclusion zones to store potential energy from light or food. You begin to lose it back to the environment and as energy is lost, inflammation rises and cells swell. The combination of all leads to molecular chaos and elevated cytokine levels.
When cytokines rise, so does inflammation and temperature. As temperature rises, oxygen tension in cells lowers because water cannot carry the energy bound in oxygen. As inflammation rises, the pH lowers as proton flows also slow and we lose our redox potential.
When a cell is oxidized, its pH is low, and pH is a measure of the strength of proton transfer to make ATP. If you cannot make ATP, your extracellular matrix, specifically, collagen begins to degrades. Collagen is hydrophilic and the key to charge separation of water. Collagen needs to be electrically induced to form a triple alpha-helix to work optimally. This is why old people get Langer lines or wrinkles in their skin when their redox potential falls. This is why older people lose their elastic ability in arteries and lungs.
When collagen loses its electric induction, it loses its water hydration shells around it. When water is lost, the ability to transfer energy is lost because water’s exclusion zone growth is limited. It reinforces a downward spiral of tissue physiology, organ function, and leads to more sedentary behavior, all of which leads to accelerated aging, increased organ failure with stress, impaired immune and vascular functions and brain aging.
This is the pathway where illness manifests first, and aging comes soon thereafter. They are inextricably linked together and this blog tells you why they are. With time, death comes knocking too early.
When non-native EMFs are also factored in as an added stressor to the mitochondrial cytochromes, the slope of decline worsens because aging tissue has more free transition metals in it that are no longer bound and water is dehydrated from collagen. This draws more electromagnetic force upon the tissues to steepen the declining slope of charge. This slope is how we should clinically be defining organ failure. Sounds hard to fathom because of the complexity, huh?
PROOF THAT THIS HAPPENS? Dean Jones at Emory University in Atlanta was the first to show that human plasma reduced glutathione/ oxidized glutathione levels is controlled at a relatively constant redox state of -137 mV in 740 healthy adults through age 50. However, after age 50, he found an oxidative shift of about 7 mV/decade occurs over the next two decades, followed by a further decline to -110 mV in the 70 to 85-year-old group.
Plasma GSH/GSSG redox in humans becomes oxidized with age, in response to chemotherapy, as a consequence of cigarette smoking, and in association with common age-related diseases like T2D, heart disease, autoimmunity, and neuro-degeneration. This implies the redox potential is tied to every neolithic disease out there we face as a species.
Cysteine’s other critical roles
While cysteine is the critical functional amino, and least abundant, of the 3 amino acids required for glutathione synthesis, it also plays key roles in other enzymes and proteins, including glutathione peroxidase 1 (GPX1). According to Cysteine-Based Redox Switches in Enzymes Antioxid Redox Signal. 2011 March: “While cysteine residues often play critical roles in enzyme catalysis, they also act as redox switches in many enzymes, allowing for communication between the global or local cellular redox properties and enzymatic function.” Cysteine is also important for forming the proper 3D structure of proteins from the disulfide bonds between cysteine groups. From Metal and redox modulation of cysteine protein function. Chem Biol. 2003 Aug:
In biological systems, the amino acid cysteine combines catalytic activity with extensive redox chemistry and unique metal-binding properties. The interdependency of these three aspects of the thiol group permits the redox regulation of proteins and metal binding, metal control of redox activity, and ligand control of metal-based enzyme catalysis. Cysteine proteins are therefore able to act as “redox switches,” to sense concentrations of oxidative stressors and unbound zinc ions in the cytosol, to provide a “storage facility” for excess metal ions, to control the activity of metalloproteins, and to take part in important regulatory and signaling pathways.
Metalloproteins work under the direction of water chemistry, the electromagnetic force, and the photoelectric status of the cell or tissues in question.
Summary
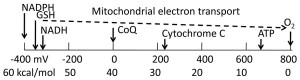
Gilbert Ling was dead right, in my opinion, that life is electronically induced. Why? The reasons are all in this blog. There is a quote from one of the references below that really says it all for the GEEKS: “The mitochondrial electron transport chain efficiently oxidizes NADH with molecular oxygen over numerous smaller steps to siphon about 98% of the energy as a proton and electrical gradient across the mitochondrial inner membrane.”
Here is the article from the National Center for Biotechnology Information.
NON GEEKS: This means that the cytochromes acts as solid-state rectifiers, or modern LEDs, or small MRI scanners, made up of proteins surrounded by hydration shells to sense small differences in electrons and protons from our environment. Semiconductor engineers call this bandgap engineering. They are able to harvest the energy and information in the subatomic particles when their charges are separated in mitochondria.
Modern science understands the inputs into mitochondria and the output of what we get from them, but how it actually happens inside the “mitochondrial black box” remains an enigma. The reason is simple. The answer is a quantum one, not a strict classically biologic one. That answer is buried in solid-state bandgap engineering: Where water, proteins, and cytochromes meet, quantum things happen and we cannot and do not perceive their occurrence.
So what does that mean to the non-geeks?
It means that the redox potential of your cells is more important than ATP. This is contrary to biochemical teachings today. Everyone in the blogosphere is pointing the problems of illness and neolithic disease at the mitochondrial level, but they have no idea of how it happens because the process is 100% tied to quantum physics. If you look at the chart above, aging and illness both have this issue in common.
The real issue is the lack of a consistent charge being maintained across the inner mitochondrial membrane and the charge on collagen all over the body to allow water to separate into positive and negative parts to transfer energy with thermodynamic freedom. It is not an ATP story at its core. This implies that all life is electronically induced. This is the core tenant of Gilbert Ling’s work and ideas.
The redox potential is a measure of the electrical potential across your membranes. The bigger the gradient is, or becomes, the more alive and well you remain. Life is a function of an organization of a cell and not its energy status. In this way, you are building a quantum machine that is metastable and is as close to a zero entropy machine there is. Water acts as a quantum motor to transfer energy to proteins and hydrophilic substances based upon its ability to charge separate in -OH groups and hydronium ions. Metastability, sleep, and a negative transmembrane potential is life’s triple play winning combo for wellness.
Moreover, these insights begin to explain why nerve action potentials are fixed stimuli and very positive in their charges. This is where protons flow and outside the nerve below the myelin is where the negatively charged DC current resides that Becker discovered. We covered a lot of this from a different viewpoint in the November 2013 webinar and the December 2013 webinar Q & A.
Energy is required in the organization to determine efficiency and wellness. When it falls below a critical level, we begin to see the organization within the quantum cell change and tissues begin to fail. These changes can be seen with MRI or in biophotons emitted from the cell. We can also see it in an altered hormone panel, which is a crude measure of the electrical and redox potential across the entire organism. It is sensitive but not specific in its ability to carry information.
An MRI, however, carries a ton of sensitivity and specificity for the problem at hand. This is why cancer cells look so different than a normal cell on MRI. Cell water in cancer is radically different in healthy and cancerous cells. Looking at a lab test can never give you that resolving power.
The redox potential is best measured by the activity of glutathione and cysteine levels in your tissues, and by how water is allowed to act in a cell to make a large exclusion zone.
What else should this redox shift imply to modern biochemists? It means that the electrons from the photoelectric effect are 98% energy efficient when mitochondria are working within the native electromagnetic force found on Earth. Nothing in biochemistry can claim this efficiency rate now. This is a perfect example of macroscopic biologic coherence published in a major journal. This contradicts everything printed in any biochemistry book not written by Gilbert Ling. His AI hypothesis said that the cells were electronically induced to animate life and metabolism.
This implies the Noble Prizes given in 1978, 1997, and 2003 should all be given to Ling at once. We all know that won’t happen, but it should. Ling was an aggressive tiger with the scientists of his day because he knew he was right. His real problem was that for his time, he was too far in front of the leading wave of biology. Whenever you find yourself on the “wrong side” of the majority, it is time to pause and reflect. Ling did not back down, and his scientific career suffered until Ray Damadian provided him refuse and took his work to create the MRI. The same thing is true of Dr. Robert O. Becker, MD’s late-career, and it is why he and Dr. Gilbert Ling have my ultimate respect. In the end, I think patients will come to realize they will never remember the words of the researchers espousing for modern cell theory, but we will remember the silence of modern healthcare, researchers, and the Nobel committee in speaking about the power buried in Ling’s work.
Ling proved them all wrong in 1952, and because he was so ahead of the curve during his time, biologists just could not figure out what he meant by electronic induction. Sometimes, being so far in front of the curve is the most dangerous place to be. Check the real-life stories about Galileo, Copernicus, or Semmelweis. Scientific dogma is nothing more than mob rule, where the majority of scientists may take away the knowledge of bright folks and hurt the public.
Wrong does not cease to be wrong because of the majority share in it. But it does rule the way science is done in this world. You need to be aware of this fallacy in modern “evidence-based medicine.”
This is why all researchers should look back to see what others before them were doing before listening to contemporaries advising them to do what they want them to do, obesity and lipid research being two great examples of this type of group think behavior going on now. Just because something isn’t a lie does not mean that it isn’t deceptive. A liar knows that he is a liar, but one who speaks mere “portions of truth” in order to deceive is a craftsman of human destruction. This is how I see the modern literature we call evidenced-based. I know these are strong words, but this blog explains, in detail, why I feel as I do.
The real problem Ling and Becker had is no one in their day had a clue how nature really worked at a subatomic level. Ling knew intuitively water acted very differently in life than it did in your sink. He just did not know precisely how. Gerald Pollack’s recent work has extended Ling’s brilliance. This is why Ling and Becker’s theories went unused for 50 odd years by medicine. The rules of quantum mechanics were being laid down when Becker and Ling were active in publishing their ideas at the tail-end of the Bell curve of the literature.
Biologists act as a group, believing that quantum mechanics has no place in biology because they believe it only defines the subatomic world. They do not realize biology and evolution are ruled by subatomic laws because the subatomic world controls DNA/RNA expression. The electromagnetic force is what “plays” our “musical instruments,” which ironically are the nucleic acids that code for Maxwell demons to animate life with the action of EZ water.
It is often stated that of all the theories proposed in this century, the silliest is quantum theory. In fact, some say that the only thing that quantum theory has going for it is that it is unquestionably correct. Biology better wise up to this. If you are a sick patient, you cannot afford to wait for them to get it right because they are going to have to rescind 3 Nobel Prizes to make this right for your health and future generations’ health. No one except Mother Nature, not even the Nobel committee, has the right to say whose idea is best or correct.
All scientists and physicians are taught to believe and think of ATP as the only high energy molecular currency a cell has. Ling was the first to show that the energy math of ATP hydrolysis could not explain the resting membrane potential, much less the Na/K ATPase that was laid on top of Peter Mitchell’s flawed chemiosmotic model. The proposed ATPase energy calculations broke the second law of thermodynamics by a 5 fold margin based upon Ling’s data! It is hard to fathom how no one saw the fallacy of this core biochemical belief based upon the negative energy balance alone.
Breaking thermodynamic rules is usually the domain of quackery but today modern biology basis all of its beliefs on this very cornerstone. It is hard to fathom when you actually understand and know the science. No one paid any attention to Ling for 55 years until this paper showed up recently. As soon I saw it in 2010, I thought of Dr. Gilbert Ling and what he has endured.
This paper clearly showed that the hydrolysis of the high energy phosphate bond produces only -7.3 kcal/mol. When you compare this value to the almost -60 kcal/mol of oxidized NADH, it becomes clear the power found in a cell is electronically induced and not driven by the hydrolysis of ATP. Electronic induction is something a quantum cell does, not one that is taught in modern cell theory’s books.
Take a look at this eye-opening table that clearly shows where the power comes from that drives all biochemical reactions. Take a look at this graph from National Center for Biotechnology Information, Copyright Exp Gerontol. 2010 March; 45(3): 173–179.)
Since the reducing energies of NADH, NADPH and GSH are all incredibly massive as seen above on the left of the picture, they can unilaterally provide the power and energy for a large number of reduction reactions in the cell including glycolysis, ATP generation, disulfide bond formation in numerous enzymes that allow for proper protein folding in the brain, all the ABC transporters, all signaling molecules and all known transcription factors. The driving force for these reactions depends on the relative concentrations of the oxidized and reduced forms of each redox couple of NADPH, NADH, and glutathione.
We need to pay deep attention to the pathways that generate these reducing intermediates and not much else. I laid the foundations of all this in the EMF 4 blog post earlier this year. The PPP is the optimal way to achieve this result. This pathway works best in cold temperatures when you are fully hydrated and primarily ketotic. The reason is simple: It exposes all your proteins to maximum water bonding sites, allowing for optimal electronic induction of all coupled biochemical reactions.
And why shouldn’t this surprise anyone? Because this is precisely how plant life does things too! The first step in photosynthesis is just this kind of charge separation and directional transfer. This is why I wrote a blog entirely devoted to photosynthesis for you before we got to this point in the Quilt. Nature uses a fractal energy plan and negative entropy plan. What plants do on their chloroplast membrane parallels what we do on our inner mitochondrial membrane.
A chlorophyll molecule, on absorbing a photon, donates an electron to an acceptor, which flows down the electron transport chain, generating ATP on the way. The ‘hole’ left behind in the donor chlorophyll is filled by an electron from another donor on the outside. That donor is water. The ‘hole’ in the case of water becomes a proton, which is also mobile, and can either accumulate in the bulk phase on one side of the membrane to create a “proton gradient” that energizes the transport of metabolites and ions across the membrane, or it can flow in a local circuit along the surface of the membrane.
The next blog gets into these flows for you and explains where they come from and how they work. Some people believe that in addition to electricity or the flow of electrons, substantial protonicity (proton electricity) also flows in the living system in the form of protons. I am one of those people.
NON-BELIEVING NON-GEEKS AND GEEKS: For all you non believers, try searching PubMed for redox or glutathione and inflammation anytime and see what you find. I got over 80,000 sites at the medical school library search I did in 2010. You will notice 80% of those all link to nuclear factor-kappa beta (NF kappa beta). It is no coincidence that NF kappa beta keeps showing up as a “redox-sensitive transcription factor” critical to immune and inflammatory response.
If you go back and re-read all my early leptin blogs from 3 years ago, you will see me constantly mention IL-6, NF kappa beta and TNF alpha. Now you might know why I began with those blogs. All of these cytokines are activate the NRF2, KEAP1, and redox pathways. The blogosphere made fun of much of what I said over three years, but who is laughing now? They are all tied to what constitutes our cellular redox potential.
The real kicker is from a cite below. Here, we see cysteine’s massive power in a quantum cell. It is the one amino acid that controls transition metal physical chemistry because of its unique disulfide bonds. The bonds interact with energy transferred to it from water charge separation. These disulfide bonds best deal with the D shell electrons that can be localized or delocalized when the electromagnetic force varies within the native environment. We get into deep trouble when the native forces are exceeded. These electrons in these metals are the ones that are used to induce charge separation (another way to get a redox potential) to power the entire cell, as Ling showed in 1952.
If cysteine levels are altered in any way, you lose your electromotive force on your inner mitochondrial membrane to move electrons from cytochrome one to reduce oxygen. You also lose the ability to use the energy in protons that are made from water and free fatty acid oxidation. It turns out that cysteine (and reduced glutathione) combines catalytic activity, to reduce the energy of activation of all biochemical reactions. Cysteine and reduced glutathione have extensive redox chemistry and unique metal-binding properties due to their exposed thiol groups. These thiols groups cannot work properly against the transition metals to contain these D shell electrons if the protein is not in the fully opened confirmation.
ATP is the molecule that controls this action of opening the protein to open water binding sites; it has nothing to do with powering any biochemical reaction. When water is fully bound to cysteine, it can transfer its energy maximally to thiol groups. This only happens when water is adjacent to a hydrophilic substance like collagen or proteins that carry a negative charge. When you eat carbs and not fat, the ATP score is not in your favor: 36 to 147, as I have pointed out many times. This difference limits the size of your battery because you cannot charge separate water. Water is life’s battery. That means you are limiting your cellular redox potential. This means you can never reach your full potential, no matter what you believe. A “paradox” is only a conflict between reality and your feeling of what reality “ought to be.”
See EMF 4, yet again, to understand why. This means cysteine, and proteins that contain this rare amino acid, act to increase energy while reducing entropy simultaneously. This is the classic definition of a “Molecular Maxwell Demon” for those following this quantum story at home. If you really want to understand what a Molecular Maxwell demon is, read Werner Loewenstein’s new book. This is the real blueprint of how evolution built a quantum cell. He too gave Peter Mitchell too much credit in his book, but Loewenstein was directionally accurate in most other points, in my view.
You only need to be directionally accurate to regain well and maintain wellness because quantum mechanics is based on probabilities not absolutes. Few “paleo research people” realize this is a fundamental principle of QED, and it is not a defect in your thinking. It all goes back to the electrons from the photoelectric effect and water chemistry in the Earth’s native environment. Energy and Epigenetics 4 gives you the parts, but how they act in unison is where wellness and life’s magic occur. Ling gave us how it begins with his Association Induction hypothesis.
QUANTUM NEURO-DEGENERATION This turns out to be a big deal in a quantum cell that is loaded with liquid crystalline semiconductors that need to be pre-loaded with electrons to remain meta-stable. Sleep keeps our semiconductors metastable. When you sleep well, you stay healthy and live longer. That the exposed thiol groups of cysteine are critically important was something Ling pointed out in the 1950s when he spoke about the disulfide bridges that form salt linkages in rigor mortis, when life dies. In rigor mortis, water is released and potassium is released, while magnesium is bound up in the disulfide salt bridges. Salts limit the size of water’s exclusion zones.
The thiol group side chain locks proteins together when energy is low or entropy is high. This is why all neuro-degenerative diseases have altered protein folding in the tertiary and quaternary configurations.
Cysteine is also important for forming the proper three dimensional structure of all proteins from the action of the disulfide bonds between cysteine groups. This is why beta-amyloid and neurofibrillary tangles are common features of all of these diseases. They are due to the action of thiol groups not fully exposed, due to low ATP levels, and this causes salt linkage binding wherever cysteine is in that protein. Exposed thiol groups surrounded by reverse water micelles (water hydration shells) allows for optimal electron withdrawing to lower the oxidation rates in all reactions in which cysteine is found.
It all comes down to what happens to the electron. This is the only charged particle used in biology that the electromagnetic force has infinite control over. In most metal proteins, you will find a transition metal not too far away from cysteine in its molecular configuration to alter its electron cloud of delocalized electrons from the D shell of these metals. This is why insulin has a zinc ion surrounded by 4 amino acid residues at its core. The interdependency of these three aspects of the thiol group permits the redox regulation of proteins and transition metal binding, transition metal control of redox activity, and ligand control of metal-based enzyme catalysis.
Cysteine proteins are therefore able to act as “Maxwell Demon redox switches” to sense the quantum particle concentrations (electrons from the photoelectric effect) of oxidative stressors and unbound zinc and aluminum ions in the cytosol, and to provide a “storage facility” for excess transition metal ions that build up in cells due to chronic oxidation of any cause, to control the activity of metalloproteins!!! This allows them to take part in important regulatory and signaling pathways.
This is why zinc and aluminum are always linked in studies on Alzheimer’s disease and Parkinson’s disease, and yet no one seems to know why. Now you do.
THE TAKE AWAY: My good friend Randy said this recently:
Many smart people through human history have denied reality because it did not match their beliefs. And we have been significantly brainwashed by media, marketing, advertising, paleo, etc. to believe what we believe about food, medicine and health. Let’s face it: to be healthy just do the opposite of what everybody believes is the truth. Fat makes you fat — so eat more fat and get skinny. Exercise burns calories so you lose weight — exercise just makes you hungrier so you eat more and gain more weight. Cold will make you sick — cold improves your immune system so embrace the cold tub. I could go on and on about what many believe is common sense, but is not. Being contrarian may not make you more friends, but may save your life.
The most powerful way to avoid aging and any neolithic disease is protect your cysteine stores, eat lots of fats and foods containing cysteine, drink lots of reverse osmosis water to bind to your opened faced protein, and avoid non-native EMF that ruins your transition metal chemistry. When they are altered, cysteine chemistry and transition metal chemistry go awry and humans die at a faster rate from all types of diseases. It is time to question all those who think they understand what is printed in a biochemistry book. Biology has imposed their beliefs to Mother Nature for far too long.
- Biology’s modern cell theory is no closer to reality than your opinion on the new Pope’s love life. Einstein’s space is no closer to reality than Van Gogh’s sky. The glory of science is not in a truth more absolute than the truth of Bach or Tolstoy, but in the act of creation itself. The scientist’s discoveries impose his own order on chaos, as the composer or painter imposes his: an order that always refers to limited aspects of reality, and is based on the observer’s frame of reference, which differs from period to period as a Rembrandt nude differs from a nude by Manet. The composer and conductor of life is the electromagnetic force. The proteins of DNA are her musical instruments.
- All in all, reality really is stranger; in fact, much stranger than we can all imagine. I particularly like the quantum idea of being positively present, absent, or negatively present simultaneously. It perplexes you until you realize QED is open to all possibilities and probabilities that allow life to adapt to all trends she may face. I have rejected modern biology’s version of “truths” because I have seen the result of that imposition in my clinic. My patients’ results are the experiments of modern biology. They have been subjected to the scientific experts’ guesses for too long. We all have seen the results of these guesses. When your guesses are added to the crossword puzzle and they are wrong, the entire puzzle becomes a chaotic mess. I think this describes where medicine is today. Mother Nature’s creatures only have biology to blame for this modern conundrum. I think it is time for her creatures to divorce biology and hitch up with QED.
Parting Shot
“I think it’s much more interesting to live and practice medicine not knowing something is correct than to have answers which might be dead wrong.” — Richard Feynman
Leave a comment.
Additional Support: Webinars by Dr. Kruse
Your Shopping List for This Post
 |
 |
 |
| N-acetyl Cysteine | Glutathione 500mg | Opti-Zinc 30mg |
 |
 |
| Physics in Mind by Werner Loewenstein | Essentia Reverse Osmosis Drinking Water |
Additional Resources
- Energy and Epigenetics 4: Light, Water, Magnetism
- Energy and Epigenetics 6: Quantum Cell Theory, Life as a Collective Phenomena
- Energy and Epigenetics 7: The Epigenetic Toolbox
- Energy and Epigenetics 8: Quantum Autism
- Energy and Epigenetics 9: Quantum Sleep
- Energy and Epigenetics 10: The Quantum Puzzle
- Energy and Epigenetics 11: Is the Force With You?
- EMF 4: Why You Might Need Carbs for Performance
- CPC #7: Obesity Qualia
- Quantum Biology 8: Quantum Scaling
- Quantum Biology 9: Photosynthesis
- Grain Brain 9: What Really Killed Michael Jackson
- The Leaky Gut Rx
- The Leptin Rx: FAQs
- The Quilt
Cites
- http://www.sciencedaily.com/releases/2013/10/131007162357.htm
- http://www.ncbi.nlm.nih.gov/pubmed/12042467
- http://www.ncbi.nlm.nih.gov/pubmed/21985671
- http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2826600/
- http://www.ncbi.nlm.nih.gov/pubmed/23664668
- http://www.ncbi.nlm.nih.gov/pubmed/12954327/ (the cysteine link)
- www.jackkruse.com/quantum-biology-9-photosynthesis/
- http://www.anti-agingfirewalls.com
- http://www.nick-lane.net/Lane-Martin%20Cell%20origin%20membrane%20bioenergetics.pdf